Exploratory analysis of diffusion tensor imaging in children with attention deficit hyperactivity disorder: evidence of abnormal white matter structure
Autor: • Giuseppe Pastura1 • Thomas Doering2 • Emerson Leandro Gasparetto3 •Paulo Mattos4 • Alexandra Prüfer Araújo1
Faça download do PDF do artigo
Received: 19 June 2015 / Accepted: 5 November 2015 / Published online: 30 November 2015
Springer-Verlag Wien 2015
ABSTRACT
Abnormalities in the white matter microstructure of the attentional system have been implicated in the aetiology of attention deficit hyperactivity disorder (ADHD). Diffusion tensor imaging (DTI) is a promising magnetic resonance imaging (MRI) technology that has increasingly been used in studies of white matter microstructure in the brain. The main objective of this work was to perform an exploratory analysis of white matter tracts in a sample of children with ADHD versus typically developing children (TDC). For this purpose, 13 drug-naive children with ADHD of both genders underwent MRI using DTI acquisition methodology and tractbased spatial statistics. The results were compared to those of a sample of 14 age- and gender-matched TDC. Lower fractional anisotropy was observed in the splenium of the corpus callosum, right superior longitudinal fasciculus, bilateral retrolenticular part of the internal capsule, bilateral inferior fronto-occipital fasciculus, left external capsule and posterior thalamic radiation (including right optic radiation). We conclude that white matter tracts in attentional and motor control systems exhibited signs of abnormal microstructure in this sample of drug-naive children with ADHD.
Keywords
Attention deficit hyperactivity disorder • Diffusion tensor imaging • White matter • Fractional anisotropy
Introduction
Attention deficit hyperactivity disorder (ADHD) describes a pattern of inattention and hyperactivity behaviours present in different settings and leads to impaired performance in social, educational and work settings (American Psychiatric Association 2013). A large number of published studies have investigated the white matter microstructure of patients with ADHD, using diffusion tensor imaging (DTI) techniques.
DTI allows the visualisation of connections between different parts of the brain, thus providing relevant information regarding integrity and connectivity (Le Bihan 2003; Mori and Zhang 2006; Silk et al. 2009a). DTI is sensitive to the direction and extent of water diffusion in the brain and uses two key measures: the mean diffusivity (MD) and fractional anisotropy (FA). MD represents the magnitude of water diffusion in any direction within the voxel and can be measured in millimetres squared per second (Silk et al. 2009a). On the other hand, FA provides information regarding the direction of water diffusion within each voxel. FA values range from 0 to 1, with 0 representing isotropic diffusion and 1 representing a predominant direction of diffusion. Whereas diffusion in the grey matter is predominately isotropic, diffusion in the white matter can be highly anisotropic (Moseley et al. 1991). As water diffusion occurs in parallel with the myelinated fibres, the FA values are higher in white matter. Increases in normal FA may represent either an increase in the density of myelinated fibres or a reduction in crossing fibres. Therefore, DTI makes it possible to examine connections between different brain areas involved in attention in patients with ADHD, whereas these connections could not be identified using conventional magnetic resonance imaging (MRI) (Conturo et al. 1999; Mori and Zhang 2006, Konrad and Eickhoff 2010; Tamm et al. 2012; Van Ewijk et al. 2012).
Previously, Ashtari et al. (2005) observed reduced FA in the right supplementary motor area, anterior portion of the right internal capsule, right cerebral peduncle, left middle cerebellar peduncle and left cerebellum in a group of patients with ADHD aged 7–11 years who were compared with typically developing children (TDC). As the supplementary motor area is related to the planning, initiation and execution of motor actions, a reduction in FA in this area is noteworthy (Amador and Fried 2004).
Using DTI, Silk et al. (2009a) observed an increase in FA in the prefrontal and temporal left hemisphere and parieto-occipital right hemisphere of patients with ADHD. According to Suzuki et al. (2003), lower neuronal branching caused by lower diffusion along the secondary and tertiary directions could explain this change. In other words, the increased FA values might indicate a reduction in neuronal branching within the pathways that conduct the fronto-temporal-parietal and occipital areas. Other previous studies (Klingberg et al. 1999; Barnea-Goraly et al. 2005; Ashtari et al. 2007) of patients with ADHD and controls corroborated this increase in FA.
corroborated this increase in FA. It is important to emphasise that Silk et al. (2009a) used tract-based spatial statistics (TBSS), a method of analysis specific to DTI studies; this method allows an analysis restricted to the centres of major white matter tracts, rather than an examination of the entire brain (Smith et al. 2006; Woolrich et al. 2009). The results reported by Silk et al. (2009a) partly corroborate the theory proposed by El-Sayed et al. (2003), whereby ADHD represents a maturational deficit that can be corrected with time.
Another study by Silk et al. (2009b) specifically investigated the basal ganglia, an important component of the fronto-striatal network that has been implicated in the pathophysiology of ADHD. In that work, the authors observed an increase in FA with age within the whole-brain volume in both the ADHD and control groups. However, in the caudate nucleus, age-related developmental changes in FA followed different pathways in the two groups. This difference might represent a delay in brain development in patients with ADHD.
The association between the brainstem and neocortex was also evaluated by Pavuluri et al. (2009). In that study, patients with ADHD and bipolar disorder were investigated using DTI, and the former group exhibited reduced FA in the anterior corona radiata, which appears to be related to fronto-striatal dysfunction.
The aim of the present study was to perform an exploratory analysis of the white matter microstructure in a sample of drug-naive children with ADHD and TDC. Given the previous findings of dysfunction in the attentional systems in studies of ADHD, we expected that children with ADHD would exhibit different FA properties.
METHODS
A sample of 13 children aged 7–10 years and of both genders was recruited at the ADHD outpatient clinic of the Federal University of Rio de Janeiro. All recruits were drug naive and met the criteria for ADHD according to the 4th edition of the Diagnostic and Statistical Manual of the American Psychiatric Association (DSM-IV) (1994). The ADHD module of the Kiddie-Schedule for Affective Disorders and Schizophrenia (K-SADS) was used in order to confirm the diagnosis (Chambers et al. 1985; Brasil and Bordin 2010). The DSM-V had not yet been published when the data were collected, but it is worth mentioning that there were no changes in the criteria and number of symptoms required for diagnosis when compared with the DSM-IV.
A control group comprising 14 age- and gender-matched TDC was recruited from the general paediatrics outpatient clinic of our university. The exclusion criteria included an intelligence quotient (IQ) of\70 and the presence of any type of non-progressive encephalopathy. IQ was estimated using the Vocabulary and Block Design subtests from the Wechsler Intelligence Scale for Children—3rd edition (Wechsler 1991; Faraone et al. 1996; Greene et al. 1996).
The only comorbidities allowed for the group of ADHD patients were other externalising conditions such as oppositional defiant disorder (ODD) and conduct disorder (CD).
MRI acquisition parameters
The patients underwent MR imaging on a 3-T system (Verio; Siemens, Erlangen, Germany), which applied standard T1- and T2-weighted sequences and an optimised DTI sequence (ecoplanar diffusion-weighted sequence along 30 gradient directions; repetition time/echo time [TR/TE] = 13,200/98, readout bandwidth = 1710 pixels/ Hz, voxel size = 2.1 mm3; no gap). The sequence parameters were as follows: T1, TR/TE = 500/8.4, 30 slices, matrix = 512 9 480, slice thickness = 4 mm and distance between slices = 5 mm; T2 flair, TR/TE = 9000/ 80, inversion time (TI) = 2500 ms, slice thickness = 4 mm, spacing between slices = 4 mm, matrix = 256 9 240; and diffusion-weighted imaging (DWI), 1b = 0 and 30b = 1000.
The Johns Hopkins University White Matter Tractography Atlas (Mori et al. 2005; Wakana et al. 2007) was used to report anatomical locations.
For voxelwise diffusion modelling, diffusion data were analysed using the FMRIB Diffusion Toolbox (http://www.fmrib.ox.ac.uk/fsl) within FSL 5.0 (Smith et al. 2004). After performing eddy current correction and brain extraction, FA images for all subjects were created by fitting a tensor model to the raw diffusion data.
A voxelwise statistical analysis of FA data was conducted using tract-based spatial statistics (TBSS) (Smith et al. 2006), a component of FSL. FA data of all subjects were aligned in a common space using the nonlinear registration tool FNIRT, which uses a b-spline representation of the registration warp field. Next, a mean FA image was created and thinned to create a mean FA skeleton representing the centres of all tracts common to the group. Each subject’s aligned FA data was then projected onto this skeleton, and the resulting data were fed into voxelwise cross-subject statistics for all voxels, with a FA cut-off of C0.20 to exclude peripheral tracts with significant intersubject variability and/or partial volume effects with grey matter. The voxelwise analysis was conducted using permutation-based inference and threshold-free cluster enhancement (TFCE), with a threshold of p\0.05 and correction for multiple comparisons (family-wise error). Corrected TFCE p value images were computed to enable the detection of differences in FA between patients and healthy controls. Every FA image was aligned to every other image to identify the most representative image, which was used as the target image. This target image was then affine-aligned into a MNI152 standard space, and every image was transformed into a 1 mm3 MNI152 space by combining the nonlinear transform to the target FA image, using the affine transform from that target to the MNI152 space. This option is recommended if it is necessary to generate a study-specific image, such as for the young children in this study.
To reduce any influence of movement that could confound the results, we visually inspected the raw diffusion Images of all subjects for head movement and excluded subjects with significant alterations. Additionally, we fixed the patients by placing cushions inside the head coil of the MR scanner to reduce any head movement. Finally, a radiologist was present throughout the exam to detect strong movements.
RESULTS
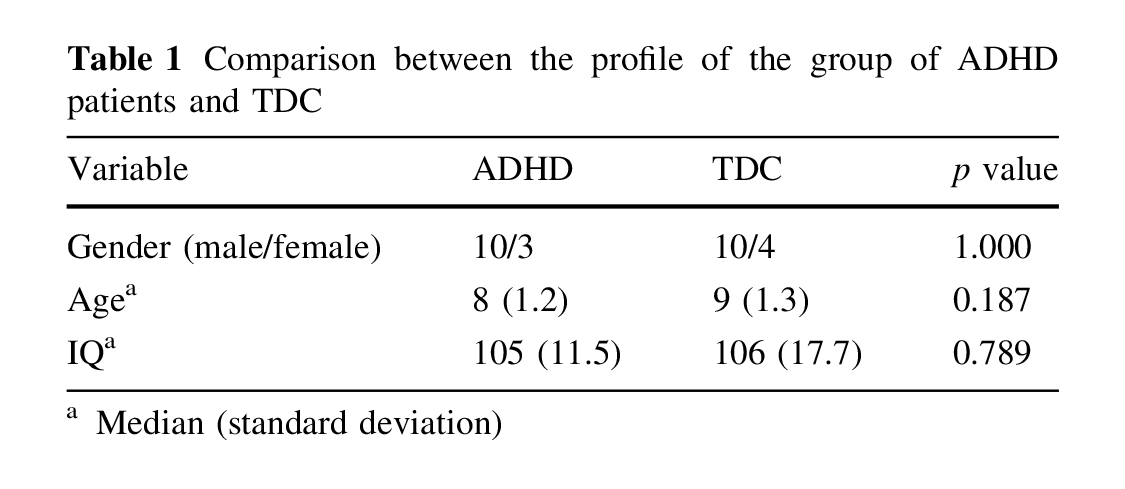
Both groups (patients with ADHD and TDC) were comparable with regard to age, gender, parental level of education and IQ (Table 1).
The most prevalent presentation of ADHD was combined, followed by inattentive and hyperactive, with frequencies of 61.6 % (n = 8), 30.7 % (n = 4) and 7.7 % (n = 1), respectively.
Psychiatric comorbidities of ADHD were present in 53.8 % (n = 7) of cases; ODD, which was present in 46.1 % (n = 6) of cases, was most prevalent, followed by CD in 15.4 % (n = 2) of cases.
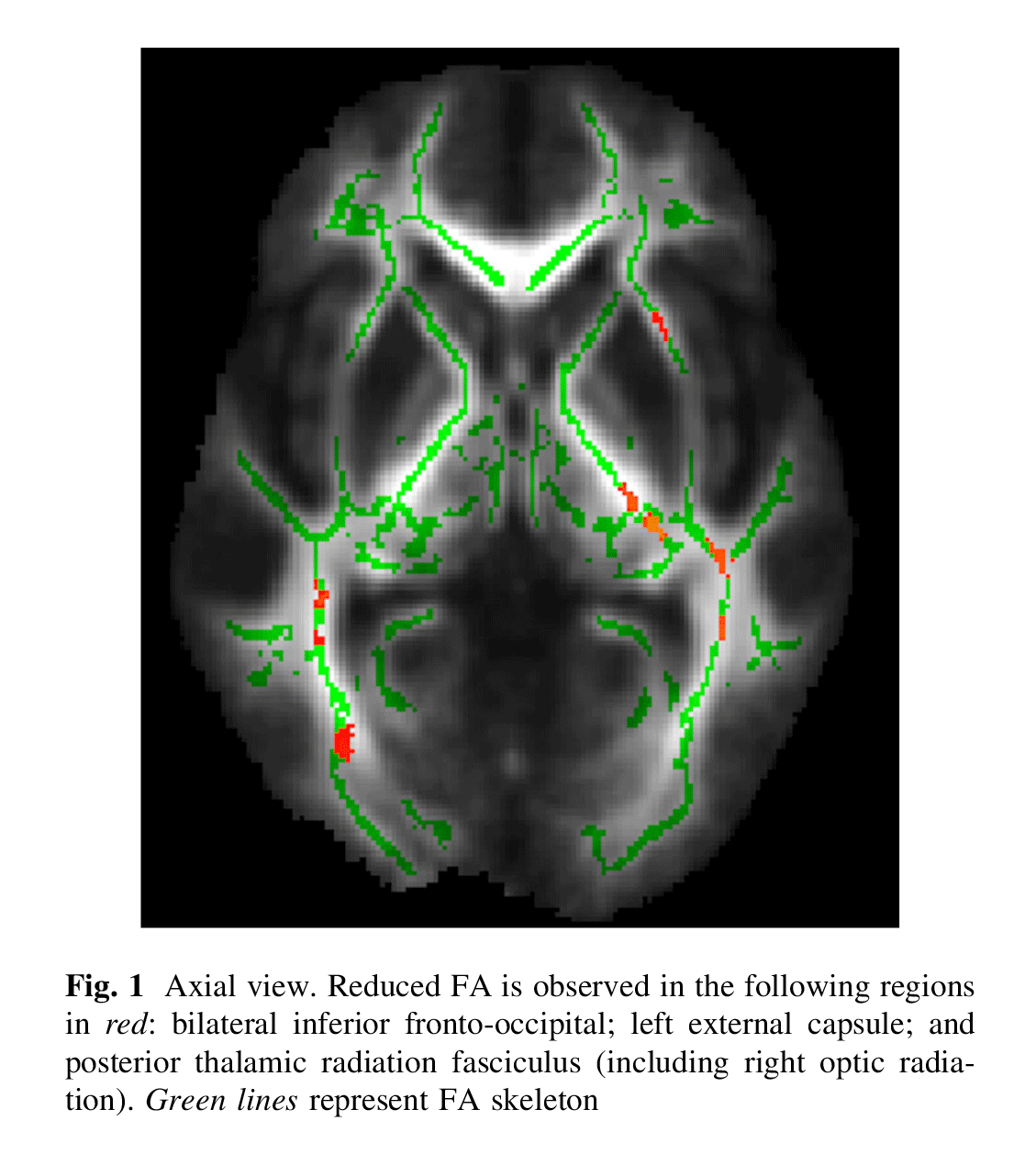
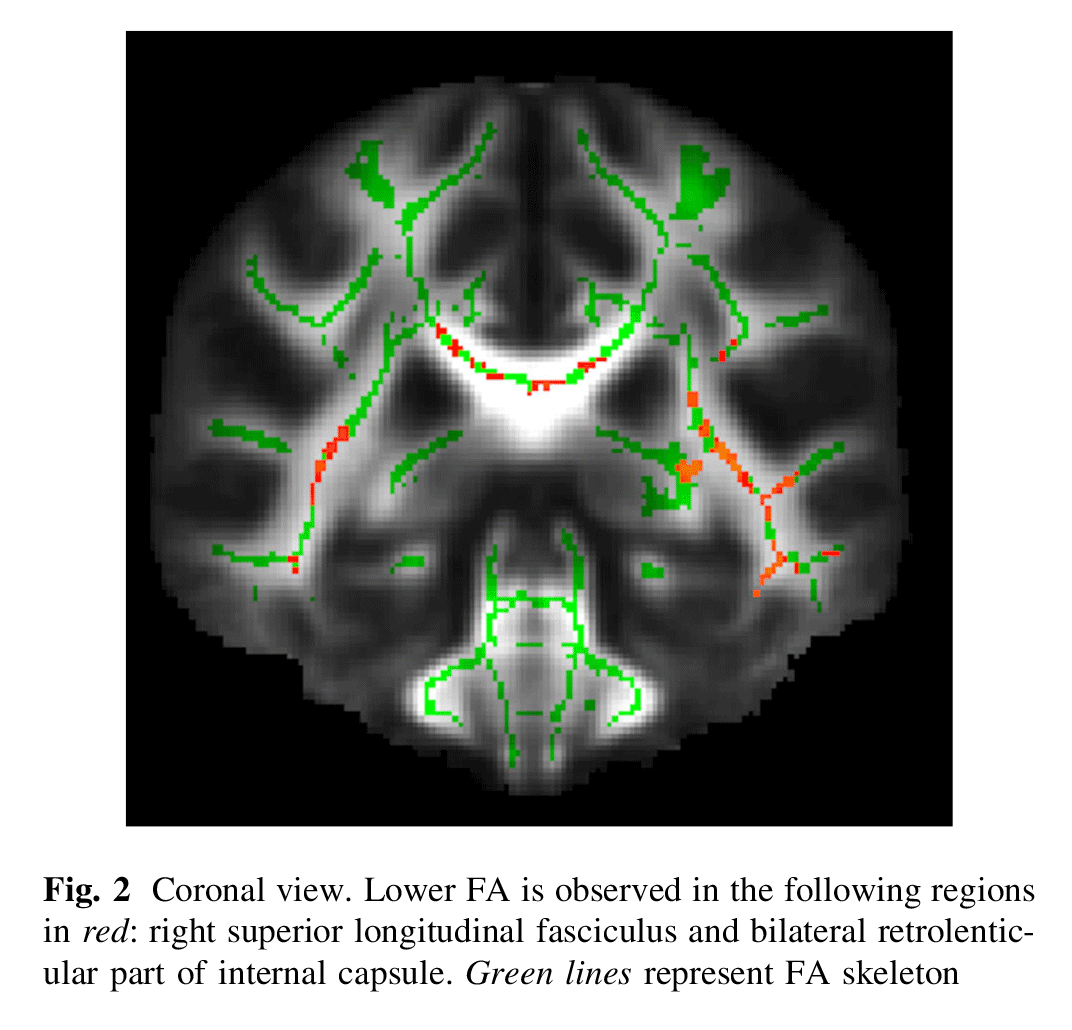
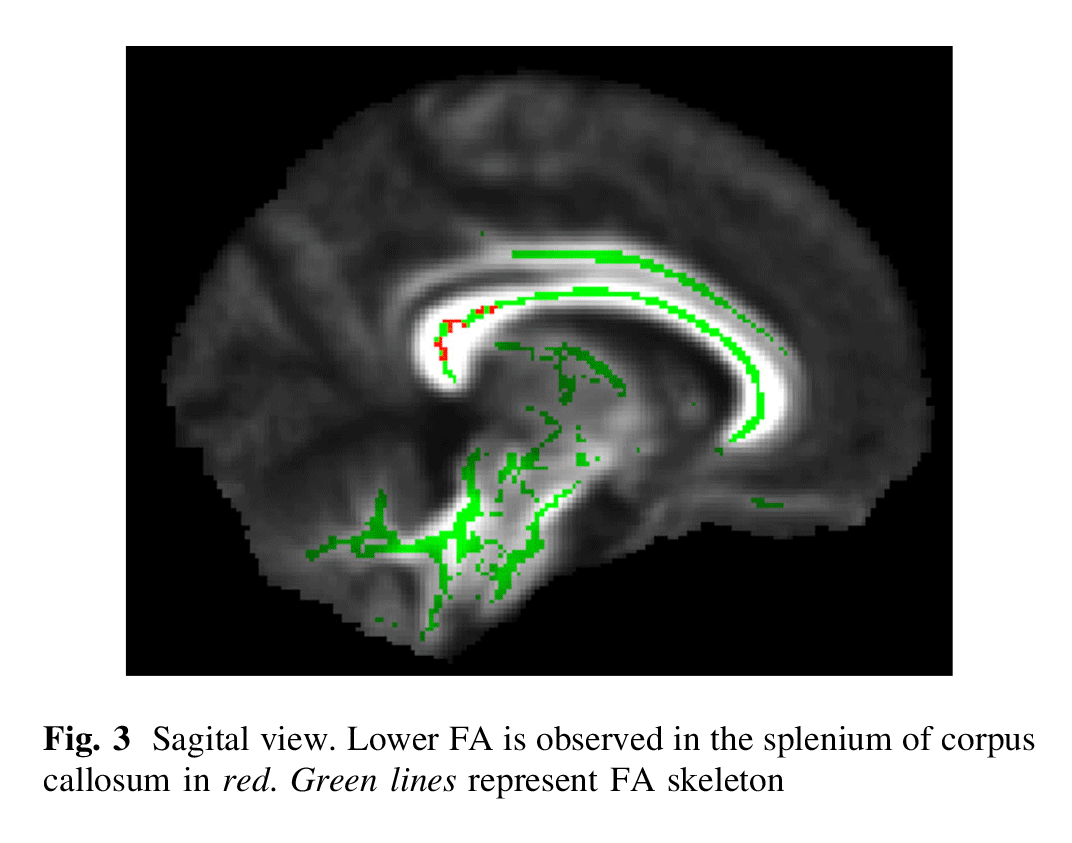
When compared with TDC, patients with ADHD had a lower FA in the following regions (Figs. 1, 2, 3): splenium of the corpus callosum, right superior longitudinal fasciculus, bilateral retrolenticular part of internal capsule, bilateral inferior fronto-occipital fasciculus, left external capsule and posterior thalamic radiation (including right optic radiation). All results survived correction for multiple testing.
DISCUSSION
In the present exploratory study, we observed reduced FA in fibres normally associated with connections between the anterior and posterior attentional systems (Bush 2010). To the best of our knowledge, this is the first study to replicate these findings in a Brazilian sample of drug-naive children with ADHD.
There is a paucity of studies concerning DTI in drugnaive children and the effects of drug treatment on the white matter microstructure in young patients with ADHD. Recently, De Luis-Garcı´a et al. (2015) published a study of DTI in patients treated with methylphenidate in which a reduction in MD was observed in medicated patients with ADHD, although no statistically significant differences were observed in FA.
Most studies concerning ADHD neurobiology address the anterior cingulate cortex, dorsolateral prefrontal cortex, prefrontal cortex and ventral-lateral parietal cortex. These areas form the cingulate-fronto-parietal attention and cognition network and are normally involved in attention, cognition, executive function, motor control, response inhibition, working memory and reward mechanisms, according to Bush (2010).
The sensory association cortices are located in the occipital, temporal and parietal lobes in the posterior region of the brain. The association cortices project sensory information to the prefrontal cortex (PFC), which regulates attention based on relevance. The PFC maintains several projections for the association cortices in order to suppress irrelevant information, sustain attention and switch attention according to task performance (Knight et al. 1989; Arnsten 2006). In a study by Mazaheri et al. (2009) involving the use of EEG during a visual attention task, patients with ADHD exhibited a disconnection between the frontal cortex, which is involved in the inhibitory control of attentional circuits, and the occipital cortex.
The corpus callosum, which is the largest white matter tract in the human brain and connects homologous areas in the cerebral hemispheres, plays a significant role in sustained attention, the division of attention between different tasks and the distribution of information between the two hemispheres under conditions of high attentional demands (Hoptman and Davidson 1994; Banich 2003; Innocenti and Bressoud 2003). The corpus callosum also operates in motor response control by transferring information to the opposite hemisphere in order to coordinate bilateral movements and inhibit contralateral motor activity during unilateral movement (Geffen et al. 1994; Barkley 1997; Bloom and Hynd 2005; Midorikawa et al. 2006). The importance of the corpus callosum in motor control is evidenced by the presence of reduced synchronisation in motor planning and the motor responses observed in patients with agenesis of the corpus callosum (Serrien et al. 2001; Caille et al. 2005).
Our results were quite distinct from those of Cao et al. (2010), whose study combined DTI and volumetry to evaluate the microstructure of the corpus callosum and its subdivisions in 28 adolescents with ADHD and 27 controls. In patients with ADHD, volumetric changes were observed in the corpus callosum as a whole, the body region and the isthmus. According to the authors, the volumetric reduction in the entire corpus callosum might reflect a reduction in the overall brain size and metabolism in patients with ADHD (Zametkin et al. 1993; Castellanos and Tannock 2002; Mostofsky et al. 2002). On the other hand, lower FA values were observed in the isthmus and these might be related to problems in the transmission of sensory information from posterior regions of the brain.
In the present study, the observation of lower FA in the right superior longitudinal fasciculus is noteworthy. This tract has important connections with the anterior cingulate, and consequently, other brain areas involved in attention such as the dorsolateral prefrontal cortex, parietal cortex, premotor cortex and striatum (Bush 2010). The anterior cingulate cortex integrates information proceeding from various sources and modulates the activity related to executive functions (Bush 2010). The anterior cingulate cortex, a component of the reward network that includes the striatum, nucleus accumbens and orbitofrontal cortex, acts by increasing decision-making efficiency (Galvan et al. 2005; Haber and Brucker 2009).
In our study, we observed a lower FA in the bilateral retrolenticular part of the internal capsule, a component of the pyramidal tract that is involved in motor control. Commonly, patients with ADHD have a comorbid developmental coordination disorder (DCD). This condition is characterised by poor motor coordination that interferes with activities of daily living and academic performance and is present in about 5 % of school-age children (Wann 2007; Zwicker et al. 2009). Symptoms of DCD persist during adolescence, and approximately 50 % of patients with DCD have comorbid ADHD (Kadesjo and Gillberg 1999; Gillberg et al. 2004; Sergeant et al. 2006; Watemberg et al. 2007). When compared with healthy children, people with DCD exhibit strong activation of the anterior cingulate cortex and weak activity between the striatum and the parietal lobe. Possibly, increased activity in the anterior cingulate cortex, a region responsible for detecting errors during task execution (Casey et al. 1997; Mathalon et al. 2003), represents compensation for the low activity in the parietal lobe (Querne et al. 2008).
Our results were similar to those of Hamilton et al. (2008), who reported reduced FA in both the corticospinal tract and the bilateral superior longitudinal fasciculus in a sample of children and adolescents with ADHD, which was compatible with deficits in motor control and attentional networks in hyperactive patients.
We emphasise that our results might have been influenced by the gender distribution in our sample. Most studies of ADHD include only males. Therefore, the inclusion of females makes our sample more representative of the population. On the other hand, it might explain some of the differences between our findings and those of the previous literature. Some studies of healthy subjects have demonstrated gender-based differences in white matter microstructure (Bava et al. 2011; Herting et al. 2012; Wang et al. 2012). Recently, King et al. (2015) showed that the white matter microstructure differs between male and female patients with ADHD. When compared to healthy controls, ADHD males exhibited lower FA in the right hemisphere regions of the corticospinal tract, inferior and superior longitudinal fasciculus and temporal portion of the superior longitudinal fasciculus, whereas females exhibited higher values in these regions. However, our small sample prevented an analysis of gender-based groups.
We must also emphasise other limitations of this study besides the small sample. We did not use intracranial volume as a covariate in our statistical analysis, although we agree that it would be a relevant parameter. Similarly, movement control and gender-specific brain volume differences were not included as confounding variables in the statistical analysis, and this might represent a limitation of the present study.
It is important to highlight limitations regarding the use of DTI in studies of the brains of individuals with neurological and psychiatric conditions. First, we must mention the absence of pre-existing normality values for white matter tracts that could serve as comparison parameters (Weyandt et al. 2013). Therefore, it becomes imperative to use a TDC sample for comparison purposes in DTI studies. In addition, the findings are not necessarily the cause of hyperactive or inattentive behaviour and often the same finding is observed in different clinical conditions (Hamilton et al. 2008; Davenport et al. 2010.). Finally, most studies involving DTI are cross-sectional, and there is no guarantee that those findings will be seen again at a subsequent time point (Weyandt et al. 2013).
Our findings demonstrate that drug-naive children with ADHD exhibit white matter microstructural anomalies in the following distinct brain networks: (a) the attentional network, which involves frontal brain regions such as the posterior thalamic radiation (including right optic radiation) and splenium of the corpus callosum; (b) networks encompassing fibres that connect anterior and posterior attentional systems such as the right superior longitudinal fasciculus, bilateral inferior fronto-occipital fasciculus and left external capsule; and (c) the corticospinal tract, which comprises the bilateral retrolenticular part of internal capsule, an area involved in motor control that is frequently altered in patients with ADHD, especially those with comorbid DCD.
References
Amador N, Fried I (2004) Single-neuron activity in the human supplementary motor area underlying preparation for action. J Neurosurg 100:250–259
American Psychiatric Association (1994) Diagnostic and statistical manual of mental disorders, 4th edition (DSM-IV). American Psychiatric Association, Washington, DC
American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders, 5th edition (DSM-V). American Psychiatric Association, Washington, DC
Arnsten AF (2006) Fundamentals of attention-deficit/hyperactivity disorder: circuits and pathways. J Clin Psychiatry 67(Suppl 8):7–12
Ashtari M, Kumra S, Bhaskar S et al (2005) Attention-deficit/ hyperactivity disorder: a preliminary diffusion tensor imaging study. Biol Psychiatry 57:448–455
Ashtari M, Cervellione KL, Hasan KM et al (2007) White matter development during late adolescence in healthy males: a crosssectional diffusion tensor imaging study. Neuroimage 35:501–510
Banich MT (2003) Interaction between the hemispheres and its implications for the processing capacity of the brain. In: Davidson RJ, Hugdahl K (eds) The asymmetrical brain. MIT Press, Cambridge, pp 261–302
Barkley RA (1997) Behavioral inhibition, sustained attention, and executive functions: constructing a unifying theory of ADHD. Psychol Bull 121:65–94
Barnea-Goraly N, Menon V, Eckert M et al (2005) White matter development during childhood and adolescence: a crosssectional diffusion tensor imaging study. Cereb Cortex 15:1848–1854
Bava Boucquey V, Goldenberg D, Thayer RE, Ward M, Jacobus J et al (2011) Sex differences in adolescent white matter architecture. Brain Res 1375:41–48
Bloom JS, Hynd GW (2005) The role of the corpus callosum in interhemispheric transfer of information: excitation or inhibition? Neuropsychol Rev 15:59–71
Brasil HH, Bordin IA (2010) Convergent validity of K-SADS-PL by comparison with CBCL in a Portuguese speaking outpatient population. BMC Psychiatry 10:83
Bush G (2010) Attention-deficit/hyperactivity disorder and attention networks. Neuropsychopharmacology 35:278–300
Caille S, Sauerwein HC, Schiavetto A et al (2005) Sensory and motor interhemispheric integration after section of different portions of the anterior corpus callosum in nonepileptic patients. Neurosurgery 57:50–59
Cao Q, Sun L, Gong G et al (2010) The macrostructural and microstructural abnormalities of corpus callosum in children with attention deficit/hyperactivity disorder: a combined morphometric and diffusion tensor MRI study. Brain Res 1310:172–180
Casey BJ, Trainor R, Giedd J et al (1997) The role of the anterior cingulated in automatic and controlled processes, a developmental neuroanatomical study. Dev Psychobiol 30:61–69
Castellanos FX, Tannock R (2002) Neuroscience of attention-deficit/ hyperactivity disorder: the search for endophenotypes. Nat Rev Neurosci 3:617–628
Chambers WJ, Puig-Antich J, Hirsch M et al (1985) The assessment of affective disorders in children and adolescents by semistructured interview: test-retest reliability of the Schedule for Affective Disorders and Schizophrenia for School-Age Children, Present Episode version. Arch Gen Psychiatry 42:696–702
Conturo TE, Lori NF, Cull TS et al (1999) Tracking neuronal fiber pathways in the living human brain. Proc Natl Acad Sci USA 96:10422–10427
Davenport ND, Karatekin C, White T, Lim KO (2010) Differential fractional anisotropy abnormalities in adolescents with ADHD or schizophrenia. Psychiatry Res 181(3):193–198
De Luis-Garcı´a R, Cabu´s-Pin˜ol G, Imaz-Roncero C, Argibay- Quin˜ones D, Barrio-Arranz G, Aja-Ferna´ndez S, Alberola-Lo´pez C (2015) Attention deficit/hyperactivity disorder and medication with stimulants in young children: a DTI study. Prog Neuropsychopharmacol Biol Psychiatry 57:176–184
El-Sayed E, Larsson JO, Persson HE et al (2003) ‘‘Maturational lag’’ hypothesis of attention deficit hyperactivity disorder: an update. Acta Paediatr 92:776–784
Faraone SV, Biederman J, Mennin D, Gershon J, Tsuang MT (1996) A prospective four-year follow-up study of children at risk for ADHD: psychiatric, neuropsychological, and psychosocial outcome. J Am Acad Child Adolesc Psychiatry 35:1449–1459
Galvan A, Hare TA, Davidson M et al (2005) The role of ventral frontostriatal circuitry in reward-based learning in humans. J Neurosci 25:8650–8656
Geffen GM, Jones DL, Geffen LB (1994) Interhemispheric control of manual motor activity. Behav Brain Res 64:131–140
Gillberg C, Gillberg IC, Rasmussen P et al (2004) Co-existing disorders in ADHD-implications for diagnosis and intervention. Eur Child Adolesc Psychiatry 13(Suppl 1):180–192
Greene RW, Biederman J, Faraone SV, Ouellette CA, Penn C, Griffin SM (1996) Toward a new psychometric definition of social disability in children with attention-deficit hyperactivity disorder. J Am Acad Child Adolesc Psychiatry 35:571–578
Haber SN, Brucker JL (2009) Cognitive and limbic circuits that are affected by deep brain stimulation. Front Biosci 14:1823–1834
Hamilton LS, Levitt JG, O’Neill J et al (2008) Reduced white matter integrity in attention-deficit hyperactivity disorder. NeuroReport 19:1705–1708
Herting MM, Maxwell EC, Irvine C, Nagel BJ (2012) The impact of sex, puberty, and hormones on white matter microstructure in adolescents. Cereb Cortex 22:1979–1992
Hoptman MJ, Davidson RJ (1994) How and why do the two cerebral hemispheres interact? Psychol Bull 116:195–219
Innocenti GM, Bressoud R (2003) Callosal axons and their development. In: Iacoboni M, Zaidel E (eds) The parallel brain: The cognitive neuroscience of the corpus callosum. MIT Press, Cambridge, pp 11–26
Kadesjo B, Gillberg C (1999) Developmental coordination disorder in Swedish 7-year-old children. J Am Acad Child Adolesc Psychiany 38:820–828
King JB, Yurgelun-Todd D, Stoeckel A, Di Muzio JM, Lopez-Larson MP (2015) Sex differences in white matter integrity in youths with attention-deficit/hyperactivity disorder: a pilot study. Front Neurosci 9:232
Klingberg T, Vaidya CJ, Gabrieli JD et al (1999) Myelination and organization of the frontal white matter in children: a diffusion tensor MRI study. NeuroReport 10:2817–2821
Knight RT, Scabini D, Woods DL (1989) Prefrontal cortex gating of auditory transmission in humans. Brain Res 504:338–342
Konrad K, Eickhoff SB (2010) Is the ADHD brain wired differently? A review on structural and functional connectivity in attention deficit hyperactivity disorder. Hum Brain Mapp 31(6):904–916
Le Bihan D (2003) Looking into the functional architecture of the brain with diffusion MRI. Nat Rev Neurosci 4:469–480
Mathalon DH, Whitfield SL, Ford JM (2003) Anatomy of an error: ERP and fMRI. Biol Psychol 64:119–141
Mazaheri A, Nieuwenhuis IL, van Dijk H, Jensen O (2009) Prestimulus alpha and mu activity predicts failure to inhibit motor responses. Hum Brain Mapp 30(6):1791–1800
Midorikawa A, Kawamura M, Takaya R (2006) A disconnection syndrome due to agenesis of the corpus callosum: disturbance of unilateral synchronization. Cortex 42:356–365
Mori S, Zhang J (2006) Principles of diffusion tensor primer imaging and its applications to basic neuroscience research. Neuron 51:527–539
Mori S, Wakana S, Nagae-Poetscher LM, Van Zijl PC (2005) MRI atlas of human white matter. Elsevier, Amsterdan
Moseley ME, Kucharczyk J, Asgari HS et al (1991) Anisotropy in diffusion weighted MRI. Magn Reson Med 19:321–326
Mostofsky SH, Cooper KL, Kates WR et al (2002) Smaller prefrontal and premotor volumes in boys with attention-deficit/hyperactivity disorder. Biol Psychiatry 52:785–794
Pavuluri MN, Yang S, Kamineni K et al (2009) Diffusion tensor imaging study of white matter fiber tracts in pediatric bipolar disorder and attention-deficit/hyperactivity disorder. Biol Psychiatry 65:586–593
Querne L, Berquin P, Vernier-Hauvette MP et al (2008) Dysfunction of the attentional brain network in children with developmental coordination disorder: a fMRI study. Brain Res 1244:89–102
Sergeant JA, Pick JP, Oosterlaan J (2006) ADHD and DCD: a relationship in need of research. Hum Mov Sci 25:76–89
Serrien DJ, Nirkko AC, Wiesendanger M (2001) Role of the corpus callosum in bimanual coordination: a comparison of patients with congenital and acquired callosal damage. Eur J Neurosci 14:1897–1905
Silk T, Vance A, Rinehart N et al (2009a) White-matter abnormalities in attention deficit hyperactivity disorder: a diffusion tensor imaging study. Hum Brain Mapp 30:2757–2765
Silk TJ, Vance A, Rinehart N et al (2009b) Structural development of the basal ganglia in attention deficit hyperactivity disorder: a diffusion tensor imaging study. Psychiatry Res 172:220–225
Smith SM, Jenkinson M, Woolrich MW, Beckmann CF et al (2004) Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage 23(S1):208–219
Smith SM, Jenkinson M, Johansen-Berg H et al (2006) Tract-based spatial statistics: Voxelwise analysis of multi-subject diffusion data. Neuroimage 31:1487–1505
Suzuki Y, Matsuzawa H, Kwee IL et al (2003) Absolute eigenvalue diffusion tensor analysis for human brain maturation. NMR Biomed 16:257–260
Tamm L, Barnea-Goraly N, Reiss AL (2012) Diffusion tensor imaging reveals white matter abnormalities in attention-deficit/ hyperactivity disorder. Psychiatry Res 202(2):150–154
Van Ewijk H, Heslenfeld DJ, Zwiers MP, Buitelaar JK, Oosterlann J (2012) Diffusion tensor imaging in attention deficit/hyperactivity disorder: a systematic review and meta-analysis. Neurosci Biobehav Rev 36(4):1093–1106
Wakana S, Caprihan A, Panzenboeck MM, Fallon JH, Perry M, Gollub RL, Hua K, Zhang J, Jiang H, Dubey P, Blitz A, van Zijl P, Mori S (2007) Reproducibility of quantitative tractography methods applied to cerebral white matter. Neuroimage 36(3):630–644
Wang Y, Adamson C, Yuan W, Altaye M, Rajagopal A, Byars AW et al (2012) Sex differences in white matter development during adolescence: a DTI study. Brain Res 1478:1–15
Wann J (2007) Current approaches to intervention in children with developmental coordination disorder. Dev Med Child Neurol 49:405
Watemberg N, Waiserberg N, Zuk L, Lerman-Sagie T (2007) Developmental coordination disorder in children with attention- deficit–hyperactivity disorder and physical therapy intervention. Dev Med Child Neurol 49:920–925
Wechsler D (1991) WISC-III/manual. The Psychological Corporation, New York
Weyandt L, Swentosky A, Gudmundsdottir BG (2013) Neuroimaging and ADHD: fMRI, PET, DTI findings, and methodological limitations. Dev Neuropsyschol 38(4):211–225
Woolrich MW, Jbabdi S, Patenaude B et al (2009) Bayesian analysis of neuroimaging data in FSL. Neuroimage 45:S173–S186
Zametkin AJ, Liebenauer LL, Fitzgerald GA et al (1993) Brain metabolism in teenagers with attention-deficit hyperactivity disorder. Arch Gen Psychiatry 50:333–340
Zwicker JG, Missiuna C, Boyd LA (2009) Neural correlates of developmental coordination disorder: a review of hypotheses. J Child Neurol 24:1273–1281
Giuseppe Pastura
giuseppe.pastura@ippmg.ufrj.br
1 Department of Pediatrics, The Federal University of Rio de
Janeiro, Rua Bruno Lobo, 50, Cidade Universita´ria,
Rio de Janeiro, RJ 21941-590, Brazil
2 CDPI Neuroimaging, Rio de Janeiro, RJ, Brazil
3 Department of Radiology, The Federal University of Rio de
Janeiro, Rio de Janeiro, RJ, Brazil
4 Department of Psychiatry, The Federal University of Rio de
Janeiro, Rio de Janeiro, RJ, Brazil